Merck Unveils First PD-1xVEGF Bispecific Data in Oncology Push
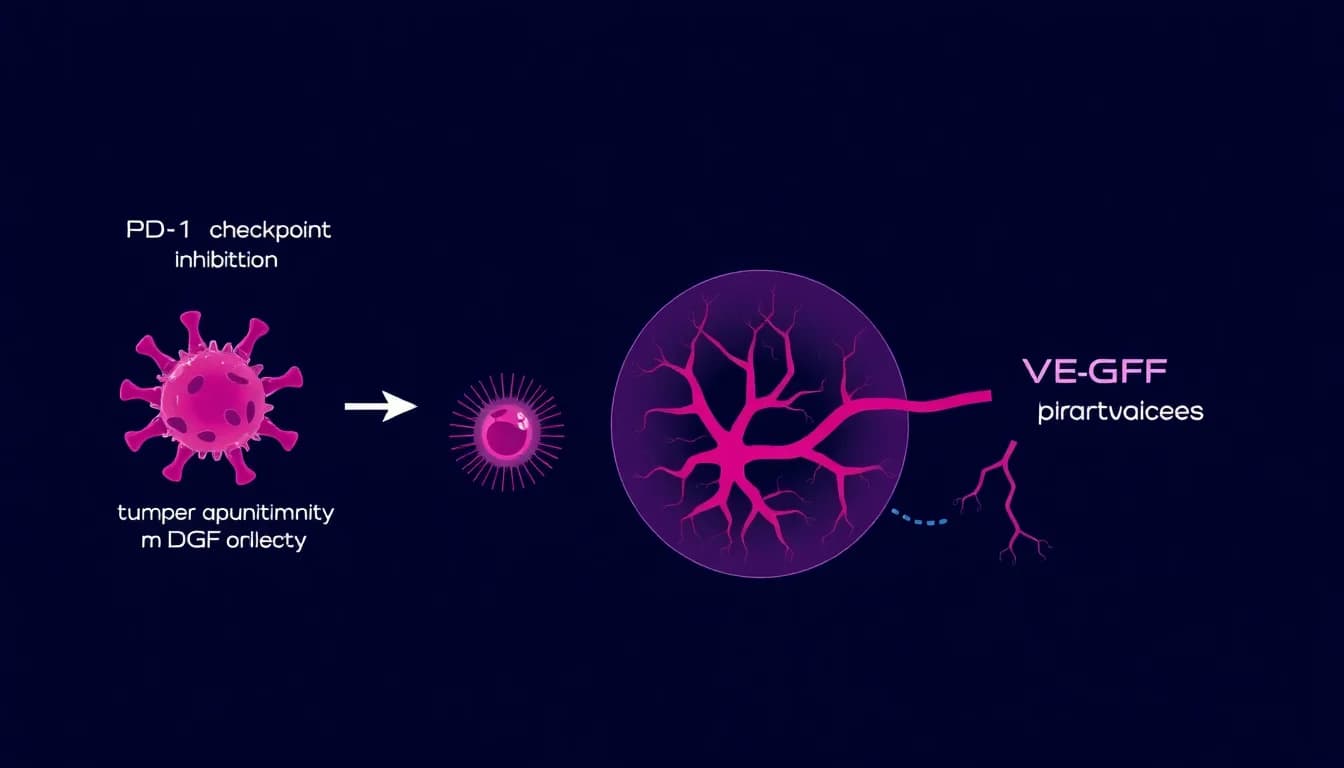
Merck & Co. has released the first clinical data from its investigational PD-1xVEGF bispecific antibody program, representing what analysts describe as a significant evolution in cancer immunotherapy strategy. The dual-mechanism approach simultaneously targets the PD-1 immune checkpoint and VEGF-mediated tumor angiogenesis, potentially addressing two critical pathways that allow tumors to evade immune detection and sustain growth.
The disclosure marks a pivotal moment in Merck's broader oncology pipeline expansion beyond its blockbuster checkpoint inhibitor Keytruda (pembrolizumab), which has dominated the PD-1 inhibitor market for nearly a decade. Industry observers note that the bispecific approach could offer advantages over combination therapy regimens that require administering separate drugs.
Dual-Mechanism Strategy: Why It Matters
The PD-1xVEGF bispecific antibody combines two well-established cancer-fighting mechanisms into a single therapeutic molecule. PD-1 (programmed cell death protein 1) inhibition has become the backbone of modern immunotherapy, helping the immune system recognize and attack cancer cells. Meanwhile, VEGF (vascular endothelial growth factor) inhibition targets the blood vessel formation that tumors require to grow and spread.
According to oncology experts, this dual approach addresses a fundamental challenge in cancer treatment: tumors often develop resistance to single-mechanism therapies. By simultaneously blocking immune evasion and cutting off nutrient supply, the bispecific design aims to create a more comprehensive anti-tumor response.
Key potential advantages of the bispecific approach include:
- Simplified dosing: One molecule instead of two separate drugs reduces administration complexity and potentially improves patient compliance
- Synergistic effects: Simultaneous pathway inhibition may produce greater efficacy than sequential or separate administration
- Improved safety profile: Potential for lower individual doses while maintaining therapeutic benefit
- Enhanced tumor penetration: Anti-angiogenic effects may improve immune cell access to tumor sites
Clinical Development Context and Early Results
While Merck has not disclosed full efficacy data in this initial presentation, the company confirmed that the Phase I/II trial enrolled patients with advanced solid tumors who had limited treatment options. The study is evaluating safety, tolerability, pharmacokinetics, and preliminary anti-tumor activity across multiple tumor types.
The bispecific antibody represents part of a broader pharmaceutical industry trend toward multi-specific therapeutics. Competitor programs from Roche, Bristol Myers Squibb, and several biotechnology companies are exploring similar dual-mechanism approaches, though each targets different pathway combinations. Industry analysts estimate the bispecific antibody market could exceed $15 billion by 2030.
For patients exploring supplement and medication interactions, the development of novel immunotherapies underscores the importance of comprehensive safety screening, as bispecific antibodies may have unique interaction profiles compared to traditional monoclonal antibodies.
Industry Reaction and Competitive Landscape
Oncology specialists responding to the data release expressed cautious optimism about the bispecific approach. Dr. Michael Chen, a medical oncologist not involved in the trial, noted that "combining checkpoint inhibition with anti-angiogenesis in a single molecule is an elegant solution to a complex problem, but we'll need to see comparative data against existing combination regimens."
The timing of Merck's disclosure is strategically significant. With Keytruda facing patent expiration in the coming years, the company has been aggressively building its next-generation oncology pipeline. The PD-1xVEGF bispecific could potentially offer differentiation in tumor types where single-agent checkpoint inhibitors have shown limited efficacy, particularly in "cold tumors" with low immune cell infiltration.
Competitors including Roche's PD-L1xTGF-β bispecific and Bristol Myers Squibb's various dual-mechanism programs are at various stages of clinical development, creating an increasingly crowded but innovative landscape in advanced cancer therapeutics.
Looking Ahead: Implications for Cancer Treatment
The path forward for Merck's bispecific program will depend heavily on upcoming Phase II expansion cohort data, expected to provide more detailed efficacy signals across specific tumor types. Regulatory experts anticipate that if early signals prove promising, the company may pursue accelerated approval pathways in selected indications where unmet medical need remains high.
For the broader oncology field, the PD-1xVEGF bispecific represents validation of the multi-specific antibody platform. Success could accelerate investment in similar dual-mechanism approaches targeting other pathway combinations, potentially reshaping how pharmaceutical companies approach cancer drug development.
Patients and healthcare providers monitoring developments in cancer immunotherapy can track emerging treatment options through resources like PharmoniQ's pharmaceutical database, which provides detailed information on both approved and investigational therapies.
As the clinical program advances, key questions remain: Will the bispecific approach demonstrate superiority over existing combination regimens? Can it overcome resistance mechanisms that limit current immunotherapies? And perhaps most importantly, will it translate to meaningful survival benefits for patients with difficult-to-treat cancers? The answers to these questions will likely emerge over the next 18-24 months as more comprehensive trial data becomes available.
Check Your Supplement Interactions
Use our AI-powered checker to analyze supplement safety and interactions.
Open Interaction Checker →Comments (0)
This article is for informational purposes only and does not constitute medical or investment advice. Content is generated with AI assistance and reviewed for accuracy.