FDA Approves Bizengri for Ultra-Rare Bile Duct Cancer Subtype
The U.S. Food and Drug Administration has granted accelerated approval to Bizengri (zenocutuzumab-zbco) for the treatment of adults with locally advanced, unresectable or metastatic cholangiocarcinoma harboring an NRG1 gene fusion. The approval, announced this week, marks the first FDA-authorized therapy specifically targeting this ultra-rare and aggressive bile duct cancer subtype, addressing a critical unmet medical need in oncology.
Bizengri becomes the seventh drug to receive approval under the National Priority Voucher Pilot program, an initiative launched to incentivize pharmaceutical development for conditions affecting extremely small patient populations. According to FDA officials, the approval demonstrates the program's effectiveness in bringing novel treatments to market for diseases that historically faced significant development challenges due to limited commercial viability.
Understanding NRG1 Fusion-Positive Cholangiocarcinoma
Cholangiocarcinoma, or bile duct cancer, already represents a rare malignancy accounting for approximately 8,000 new diagnoses annually in the United States. Within this already limited population, NRG1 gene fusions occur in an estimated 1-3% of cases, making this particular subtype exceptionally uncommon. Patients with NRG1 fusion-positive disease have historically had extremely limited therapeutic options beyond standard chemotherapy regimens.
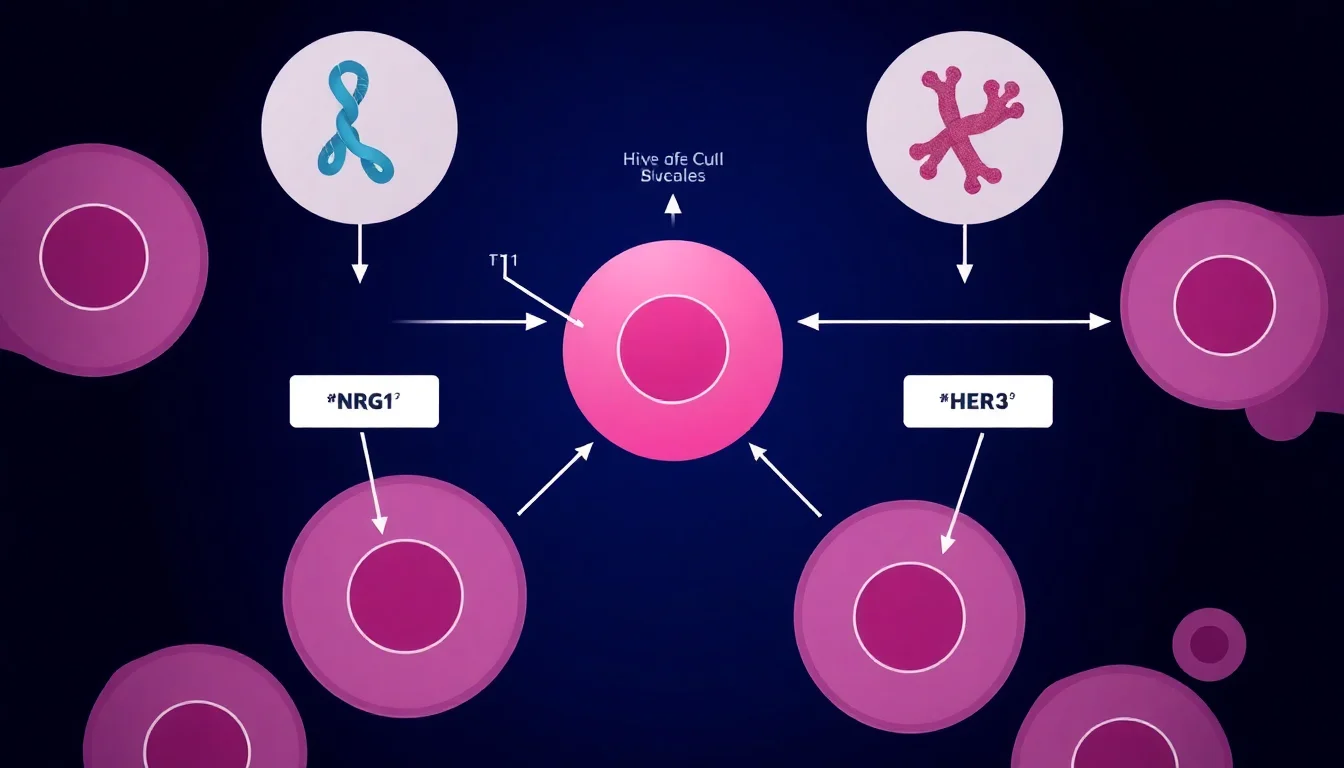
NRG1 gene fusions result in abnormal protein signaling that drives cancer cell growth and survival. Bizengri works as a bispecific antibody designed to bind simultaneously to NRG1 and its receptor HER3, effectively blocking the growth signal that fuels tumor progression. This targeted mechanism represents a significant advancement over conventional cytotoxic approaches.
Clinical Evidence Supporting Approval
The FDA's accelerated approval was based on compelling data from a multicenter clinical trial evaluating Bizengri in patients with NRG1 fusion-positive solid tumors, including cholangiocarcinoma. Key findings included:
- Overall response rate: Approximately 40% of cholangiocarcinoma patients demonstrated tumor shrinkage
- Duration of response: The majority of responders maintained their response for six months or longer
- Safety profile: Most common adverse events included infusion-related reactions, diarrhea, and musculoskeletal pain, generally manageable with supportive care
- Biomarker-driven approach: Patients were selected based on confirmed NRG1 fusion status through comprehensive genomic profiling
The trial design reflects the growing importance of precision oncology, where treatment selection is guided by specific molecular alterations rather than tumor location alone. Diagnostic testing for NRG1 fusions will be essential for identifying appropriate candidates for Bizengri therapy.
Priority Voucher Program Impact
Industry analysts note that Bizengri's approval underscores the National Priority Voucher Pilot program's role in addressing pharmaceutical development gaps for ultra-rare diseases. The program, established to encourage innovation in areas with extremely limited patient populations, provides regulatory incentives and streamlined pathways for qualifying therapies.
"This approval validates the Priority Voucher framework as a mechanism for bringing meaningful treatments to patients who would otherwise have virtually no options," according to pharmaceutical policy experts familiar with the program. The initiative has now facilitated seven approvals since its inception, covering diverse therapeutic areas where traditional development economics might discourage investment.
For patients with cholangiocarcinoma, access to comprehensive genomic testing becomes increasingly critical. Healthcare providers should consider molecular profiling tools that can identify actionable biomarkers like NRG1 fusions, enabling appropriate treatment selection and potential clinical trial enrollment.
Looking Ahead: Implications for Precision Oncology
Bizengri's approval signals several important trends for the pharmaceutical industry and cancer care. First, it reinforces the viability of developing therapies for molecularly defined patient subsets, even when the absolute numbers are extremely small. This precision medicine approach is becoming standard practice across oncology, with complementary approaches also being explored in supportive care contexts.
Second, the accelerated approval pathway requires continued clinical study to verify and describe the drug's clinical benefit. The manufacturer will need to conduct confirmatory trials, and the FDA retains authority to withdraw approval if subsequent studies fail to demonstrate meaningful patient outcomes.
Finally, this development highlights the ongoing need for improved diagnostic accessibility. Many community oncology practices lack routine access to comprehensive genomic profiling that can detect rare fusions like NRG1. Expanding testing infrastructure and reimbursement will be essential to ensure eligible patients can benefit from targeted therapies like Bizengri.
For pharmaceutical companies, the approval demonstrates that focused development in ultra-rare indications can succeed with appropriate regulatory support and robust clinical evidence. As precision oncology continues advancing, similar biomarker-driven approvals are expected across multiple cancer types, fundamentally changing how treatments are developed and prescribed.
Check Your Supplement Interactions
Use our AI-powered checker to analyze supplement safety and interactions.
Open Interaction Checker →Comments (0)
This article is for informational purposes only and does not constitute medical or investment advice. Content is generated with AI assistance and reviewed for accuracy.